Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Home Learning Center Glycerine
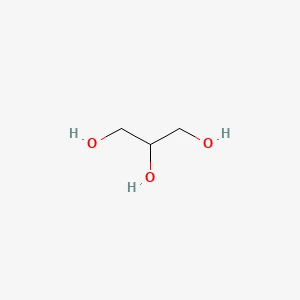
Glycerine Structure
| IUPAC Name | Propane-1,2,3-triol |
| CAS Number | 56-81-5 |
| HS Code | 2905.45 |
| Molecular Formula | C3H8O3 |
| Structural Formula | HOCH2-CH(OH)-CH2OH |
| Synonyms | Glycerol, Glycerin, 1,2,3-Propanetriol, Trihydroxypropane |
| Molecular Weight | 92.09 g/mol |
Glycerine (glycerol) is a simple polyol compound with three hydroxyl groups. It is a colorless, odorless, viscous liquid that is sweet-tasting and non-toxic. As a byproduct of biodiesel production and fat hydrolysis (saponification), glycerine is abundantly available as a co-product of oleochemical manufacturing.
Glycerine is highly hygroscopic and miscible with water and alcohols, which explains its extensive use as a humectant in food, pharmaceutical, and personal care applications.
Industrially, glycerine is used in a vast range of products including pharmaceuticals (cough syrups, suppositories), food (sweetener, humectant, solvent), personal care (moisturizer, soap), and as a chemical intermediate for synthesizing nitroglycerine, alkyd resins, and more.
| Physical Description | Clear, colorless, viscous liquid. |
| Color / Form | Colorless. |
| Odor | Essentially odorless. |
| Taste | Sweet. |
| Boiling Point | 290 °C (decomposes partially) |
| Melting Point | 17.8 °C |
| Flash Point | 160 °C |
| Solubility | Completely miscible with water and ethanol; insoluble in most oils. |
| Density | 1.261 g/cm³ |
| Vapor Density | Greater than air. |
| Vapor Pressure | Very low at ambient temperature. |
| Stability / Shelf Life | Stable under normal conditions. Hygroscopic; absorbs moisture from the air. |
| Viscosity | Highly viscous liquid (~1490 mPa·s at 20 °C). |
| Heat of Combustion | Approx. −1,655 kJ/mol. |
| Polymerization | No hazardous polymerization. |
| Ionization Potential | No data available. |
Glycerine is one of the most versatile chemicals in commercial use, with applications spanning food, pharmaceutical, personal care, and industrial sectors. In food applications, it is used as a humectant (E422) in soft foods, baked goods, and confectionery to prevent drying and improve shelf life. It functions as a solvent for food flavors and colors, a sweetener in sugar-free products, and a viscosity modifier in beverages and syrups.
In pharmaceuticals, glycerine is used in a remarkably wide range of formulations. It serves as a solvent and co-solvent for active ingredients, a humectant in topical formulations, a lubricant in suppositories and enemas, a component of cough mixtures and throat sprays, and a plasticizer for gelatin capsules. Glycerol is also used in intravenous formulations for treating cerebral edema.
In personal care and cosmetics, glycerine is the most widely used humectant and moisturizer in skin care products, providing hydration by attracting moisture from the environment. It is a key ingredient in hand creams, body lotions, liquid soaps, shaving preparations, toothpastes, and makeup formulations.
Major industrial uses include synthesis of nitroglycerine (for explosives and pharmaceuticals), production of alkyd resins for paints and coatings, polyurethane foam production, and antifreeze formulations. Glycerine is also used as a component in electronic cigarette (e-cigarette) liquids, as a smoke fluid in theatrical fog machines, and as a medium for preserving biological specimens.
Glycerine is primarily obtained as a co-product of two major industrial processes: transesterification (biodiesel production) and saponification (soap making).
In biodiesel production, vegetable oils or animal fats react with methanol in the presence of a base catalyst (NaOH or KOH) to produce fatty acid methyl esters (biodiesel) and crude glycerine as a 10% co-product by weight. Crude glycerine contains water, methanol, soap, and inorganic salts and must be purified.
In oleochemical fatty acid production, fat splitting (steam hydrolysis) yields a glycerine-water mixture (sweet water) as a co-product. This is concentrated and further processed similarly to biodiesel-derived glycerine.
Purification of crude glycerine involves neutralization (to remove soap and convert fatty acids), distillation (to remove water and methanol), activated carbon treatment (for decolorization), and ion exchange (for desalting). Refined glycerine is produced in USP/food grade (99.5%+ purity) and technical grade (95%+ purity) forms.
Low toxicity. Generally regarded as safe (GRAS) for food use. May cause mild eye irritation on direct contact.
Combustible liquid. Flash point is relatively high (160 °C).
Non-irritating under normal conditions. Eye contact with pure glycerine may cause temporary discomfort due to osmotic effect.
Combustible liquid with relatively high flash point.
Rinse with water if desired; generally well tolerated.
Rinse eyes with water if contact occurs.
Generally safe in food quantities. Seek advice if very large amounts are ingested.
Use water spray, foam, or CO₂. Glycerine fires may be difficult to extinguish; use large volumes of water.
Small spill: Absorb with inert absorbent material (sand, cloth, paper). Collect in labeled containers. Rinse area with water. Prevent large quantities from entering drains (causes BOD load in waterways).
Large spill: Contain with bunds. Pump or scoop into recovery containers. Dilute small residual quantities with water before draining. Report significant spills to environmental authorities.
Store in original, tightly closed containers in a cool, dry, well-ventilated area. Glycerine is highly hygroscopic — keep containers sealed to prevent absorption of atmospheric moisture, which dilutes the product and promotes microbial growth. Keep away from strong oxidizing agents (e.g., chromic acid, potassium permanganate, nitric acid), with which it can react violently. Store away from heat sources and open flames.
Recommended storage temperature: 15–25 °C. Shelf life: 24 months in properly sealed containers. For pharmaceutical and food grade: store in food-grade HDPE, stainless steel 304/316, or glass containers. Avoid aluminum and iron which can be corroded by hot glycerine. Prevent moisture ingress. Long-term bulk storage tanks should be sealed and nitrogen-blanketed.
For more detailed information including pricing, customization, and shipping:
Discover more about this product and understand how it may fit into your operations, formulations, or production requirements.
Browse Product