Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Home Learning Center Lauryl Alcohol
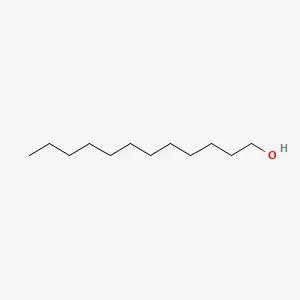
Lauryl Alcohol Structure
| IUPAC Name | Dodecan-1-ol |
| CAS Number | 112-53-8 |
| HS Code | 2905.16 |
| Molecular Formula | C12H26O |
| Structural Formula | CH3(CH2)10CH2OH |
| Synonyms | 1-Dodecanol, n-Dodecyl alcohol, C12 fatty alcohol, Dodecanol |
| Molecular Weight | 186.33 g/mol |
Lauryl alcohol (1-dodecanol) is a saturated fatty alcohol with a 12-carbon chain. It is an important oleochemical intermediate derived from the reduction of lauric acid or from petrochemical sources.
Lauryl alcohol is a key precursor for the synthesis of sodium lauryl sulfate (SLS), sodium laureth sulfate (SLES), and other anionic surfactants that are among the most widely used detergent and foaming agents in personal care products.
Beyond surfactant production, lauryl alcohol is used as a flavoring agent, in personal care formulations as an emollient, and as a plasticizer and lubricant in industrial applications.
| Physical Description | White crystalline solid or colorless liquid (above melting point) with a mild fatty odor. |
| Color / Form | White solid; colorless liquid when melted. |
| Odor | Mild fatty, characteristic odor. |
| Taste | Mild, fatty. |
| Boiling Point | Approx. 263 °C |
| Melting Point | 24–26 °C |
| Flash Point | ~127 °C |
| Solubility | Practically insoluble in water; soluble in ethanol, ether, and most organic solvents. |
| Density | Approx. 0.831 g/cm³ |
| Vapor Density | Greater than air. |
| Vapor Pressure | Low at ambient temperature. |
| Stability / Shelf Life | Stable under normal conditions. |
| Viscosity | Low viscosity liquid above melting point. |
| Heat of Combustion | Approx. −7,450 kJ/mol. |
| Polymerization | No hazardous polymerization. |
| Ionization Potential | No data available. |
Lauryl alcohol is the most important C12 fatty alcohol commercially, primarily due to its role as the key raw material for the manufacture of sodium lauryl sulfate (SLS) and sodium lauryl ether sulfate (SLES), which together are the backbone surfactants in the majority of personal care cleansing products.
In the production of SLS, lauryl alcohol is sulfated using SO₃ or chlorosulfonic acid, then neutralized with NaOH to yield the sodium salt. For SLES, lauryl alcohol is first ethoxylated (reaction with 1–3 moles of ethylene oxide under pressure and base catalysis) to produce laureth-1 to laureth-3, which are then sulfated and neutralized.
In the fragrance industry, lauryl alcohol is oxidized to lauraldehyde (dodecanal), a fragrance ingredient with a sweet, floral, citrus-like odor used in perfumery and as a flavor component. Lauryl acetate and other lauryl esters are also used in flavors and fragrances.
Industrial uses include application as a plasticizer for cellulose and vinyl resins, a defoaming agent in papermaking and fermentation, a carrier in agricultural adjuvant formulations, and a component in specialty lubricants and metalworking fluids.
Non-ionic surfactants (lauryl alcohol ethoxylates, C12 AEO) produced from lauryl alcohol are used in laundry detergents, dish care products, industrial cleaners, and emulsifier blends for food, cosmetics, and agriculture.
Lauryl alcohol (1-dodecanol) is produced commercially by catalytic hydrogenation of methyl laurate (methyl dodecanoate) or lauric acid under high-pressure hydrogen in the presence of copper chromite or copper-zinc catalyst at temperatures of 200–280 °C and hydrogen pressures of 200–300 bar.
The crude fatty alcohol product is purified by fractional distillation under vacuum to yield a high-purity C12 fraction. An alternative synthetic route is the Ziegler process (oligomerization of ethylene followed by oxidation and hydrolysis), which provides a linear C12 fatty alcohol from petrochemical feedstock. This route is used by some large-scale chemical manufacturers, though oleochemical routes are now preferred.
Quality parameters include GC purity (>98% for technical grade, >99.5% for cosmetic grade), hydroxyl value, acid value, iodine value, color (APHA), and moisture.
May cause mild skin and eye irritation on direct contact. Low acute systemic toxicity.
Combustible. Flash point above ambient temperature (~127 °C).
Mild skin and eye irritant. May cause drying of skin on repeated contact.
Combustible liquid/solid.
Wash with soap and water.
Flush with water for at least 15 minutes.
Rinse mouth. Seek medical advice.
Use CO₂, foam, or dry chemical.
Small spill: Absorb with inert material (sand, dry earth). Collect in labeled, sealable containers. Ventilate area. Prevent entry into drains.
Large spill: Contain with bunds or earth. Do not allow entry into sewers or waterways. Pump into recovery containers. Ventilate area to disperse vapors. Dispose of collected material according to local regulations.
Store in original, tightly closed containers in a cool, dry, well-ventilated area. Keep away from heat sources, open flames, oxidizing agents, and strong acids. Ground and bond containers and equipment during transfer to prevent static buildup. Maintain good ventilation in storage area. Inspect containers regularly for integrity.
Recommended storage temperature: 25–35 °C (just above melting point of 24–26 °C) for liquid handling; 15–25 °C for solid storage. Shelf life: 24 months in original sealed containers. Suitable containers: stainless steel 304/316, HDPE, or mild steel (uncoated acceptable for short-term). Protect from moisture and oxidizing agents. Nitrogen blanket for bulk liquid storage.
For more detailed information including pricing, customization, and shipping:
Discover more about this product and understand how it may fit into your operations, formulations, or production requirements.
Browse Product