Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Home Learning Center Methyl Caproate
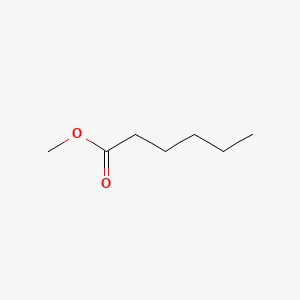
Methyl Caproate Structure
| IUPAC Name | Methyl hexanoate |
| CAS Number | 106-70-7 |
| HS Code | 2915.90 |
| Molecular Formula | C7H14O2 |
| Structural Formula | CH3(CH2)4COOCH3 |
| Synonyms | Methyl hexanoate, Hexanoic acid methyl ester, Caproic acid methyl ester, C6:0 methyl ester |
| Molecular Weight | 130.19 g/mol |
Methyl caproate is the methyl ester of caproic acid (hexanoic acid, C6:0). It is one of the lighter medium-chain fatty acid methyl esters obtained from the fractionation of coconut or palm kernel oil methyl esters.
Methyl caproate has a characteristic fruity, cheese-like odor and is used as a flavoring agent and fragrance component. It is also an important intermediate in the oleochemical industry and a component of coconut-derived biodiesel.
Its relatively low boiling point and high solubility in organic solvents make it useful as a solvent and process chemical.
| Physical Description | Clear, colorless, mobile liquid. |
| Color / Form | Colorless. |
| Odor | Fruity, cheese-like, fatty odor. |
| Taste | Fruity, slightly rancid. |
| Boiling Point | Approx. 150 °C |
| Melting Point | -71 °C |
| Flash Point | ~49 °C |
| Solubility | Slightly soluble in water; miscible with ethanol and organic solvents. |
| Density | Approx. 0.884 g/cm³ |
| Vapor Density | Greater than air. |
| Vapor Pressure | Moderate at ambient temperature. |
| Stability / Shelf Life | Stable under normal conditions. Flammable; keep away from ignition sources. |
| Viscosity | Very low viscosity (mobile liquid). |
| Heat of Combustion | Approx. −3,900 kJ/mol (estimated). |
| Polymerization | No hazardous polymerization. |
| Ionization Potential | No data available. |
Methyl caproate is used extensively in the flavor and fragrance industry due to its fruity, fermented, cheese-like odor character. It is a component of pineapple, banana, apple, and dairy flavor compositions, and is also used to create cognac, wine, and rum-like flavor notes in food and beverage applications. As a fragrance ingredient, it is used in concentrations below 0.1% to add fatty, fruity freshness to floral and fruity fragrance compositions.
As a chemical intermediate, methyl caproate can be hydrogenated to produce 1-hexanol (hexyl alcohol), used in flavors and fragrances, or used in synthesis of hexyl esters. It can also be reduced to hexanal, a green, fatty-smelling aldehyde with applications in fine fragrance.
As a component of coconut oil-based biodiesel (COME), methyl caproate is present at low levels (~0.5–1%) and contributes to the very low cloud point of coconut biodiesel relative to palm or soybean biodiesel.
Industrial solvent applications include use as a specialty solvent for natural resins, extraction solvent in food-grade processes, and as a carrier/diluent in specialty coatings and inks requiring rapid evaporation. In research settings, it is used as an analytical reference standard for GC FAME analysis methods.
Methyl caproate is produced by transesterification of caproic acid-containing oils (principally coconut oil or palm kernel oil, which contain ~0.5–0.8% C6 fatty acids) with methanol, or by direct esterification of caproic acid with methanol using acid catalysis.
Due to its relatively low boiling point (150 °C) compared to other FAME components, methyl caproate is easily isolated from crude FAME mixtures by fractional distillation at near atmospheric pressure or under mild vacuum. The separation from adjacent methyl caprylate (C8, bp ~193 °C) is straightforward.
High-purity methyl caproate (>99% GC) for flavor and fragrance use requires careful redistillation to remove trace impurities that can affect odor quality. Quality parameters include GC purity, acid value, refractive index, density, and FEMA/EFFA flavor safety certification for food-grade material.
Flammable liquid. May cause skin and eye irritation. Vapor may cause irritation of respiratory tract at elevated concentrations.
Flammable liquid (flash point ~49 °C). Keep away from heat, sparks, and open flames.
Skin, eye, and respiratory irritant.
Flammable liquid — flash point below 60 °C. Vapors are heavier than air and may accumulate.
Wash with soap and water. Remove contaminated clothing.
Flush with water for 15 minutes. Seek medical attention.
Do not induce vomiting. Seek medical attention.
Use CO₂, dry chemical, or foam. Do not use water jet.
FLAMMABLE — eliminate all ignition sources immediately.
Small spill: Absorb with dry sand or non-combustible absorbent. Collect in sealed containers. Do not use water jet which may spread flammable liquid. Ventilate area thoroughly.
Large spill: Evacuate area. Contain with bunds. Keep away from drains. Use explosion-proof equipment for recovery. Ventilate heavily. Notify fire brigade and environmental authorities.
FLAMMABLE — store away from all ignition sources including electrical equipment, open flames, and heat. Store in original, tightly closed containers in a cool, well-ventilated area or dedicated flammable liquid storage room. Use explosion-proof electrical fittings in storage area. Ground and bond containers during transfer. Keep away from oxidizing agents, strong acids, and strong bases.
Recommended storage temperature: 10–20 °C. Do not store above 30 °C for extended periods. Flash point ~49 °C — comply with local regulations for flammable liquid storage. Shelf life: 24 months in original sealed containers. Suitable containers: stainless steel 304/316, HDPE, or aluminum. Nitrogen blanket recommended. Store in dedicated flammable materials store. Containers must be kept sealed — vapor pressure is moderate at ambient temperature.
For more detailed information including pricing, customization, and shipping:
Discover more about this product and understand how it may fit into your operations, formulations, or production requirements.
Browse Product